Ethylene Sulfate: Properties, Uses and Safety Guide
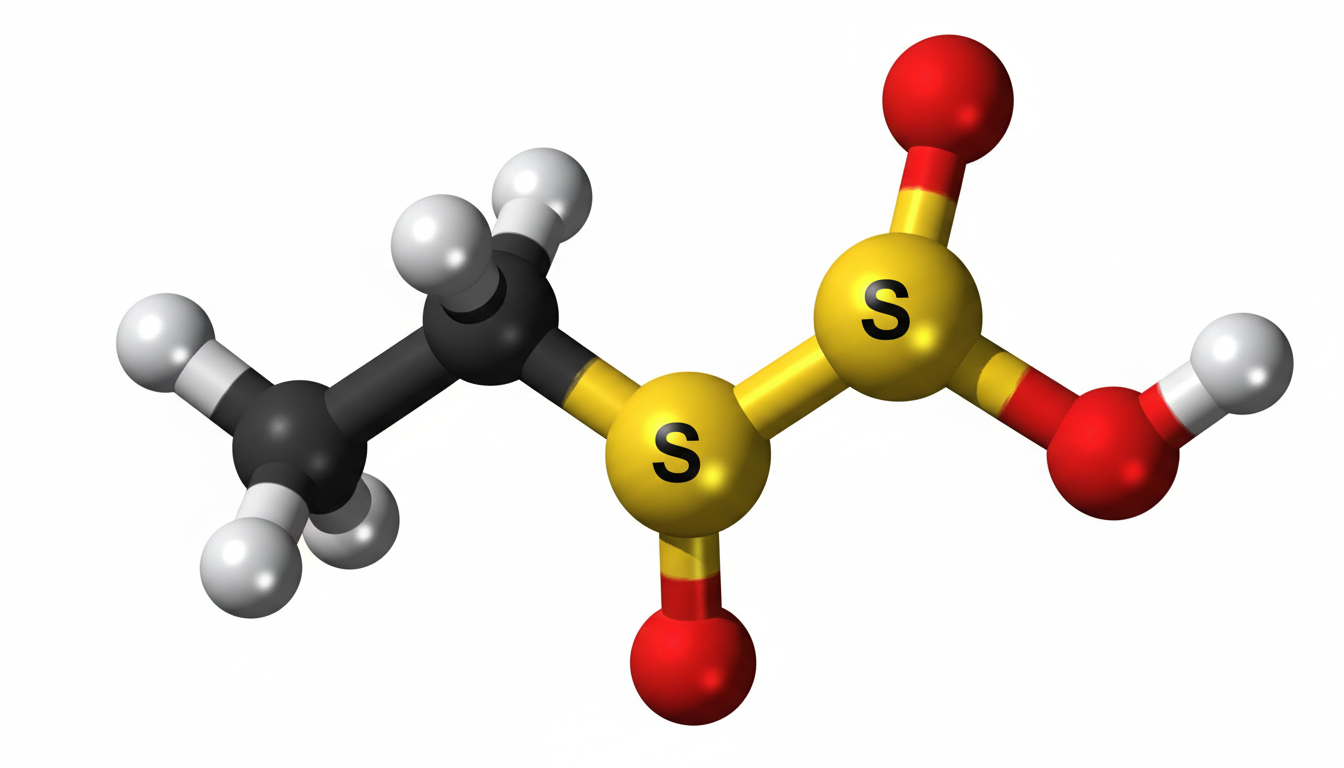
Ethylene sulfate is an important chemical compound that is widely studied in industrial chemistry due to its unique structure and reactive nature. It is often associated with organic synthesis processes and plays a role in the development of various chemical intermediates. In many industrial applications, ethylene sulfate is valued for its ability to participate in controlled reactions, making it useful in specialized manufacturing processes. Understanding its characteristics helps researchers and industries utilize it safely and efficiently.
Ethylene sulfate is typically handled in controlled environments because of its chemical reactivity. It is not a household chemical and is mainly used in laboratories and industrial facilities. Its significance lies in its role as an intermediate compound, helping to produce more complex substances used in different sectors such as materials science and chemical engineering.
Chemical Structure and Composition
The structure of ethylene sulfate is based on an ethylene backbone combined with sulfate groups, which gives it distinct chemical behavior. This arrangement allows it to participate in reactions that involve ring-opening and nucleophilic attack, which are common in organic chemistry processes.
The molecular configuration makes it a highly reactive compound under specific conditions. Because of this, it is often stored under carefully controlled temperatures and environments to prevent unwanted decomposition or reaction. Its structural properties also make it a useful intermediate in synthesis reactions where precision and stability are required.
Physical and Chemical Properties
Ethylene sulfate exhibits properties that are important for its handling and application. It generally appears as a stable compound under standard laboratory conditions but can react when exposed to moisture, heat, or strong reactive agents.
One of its key chemical properties is its ability to act as an electrophilic compound, meaning it can accept electrons during chemical reactions. This makes it valuable in forming new chemical bonds. Its stability in controlled environments allows chemists to use it in planned reactions without rapid degradation.
From a physical standpoint, ethylene sulfate is typically managed in sealed containers to avoid exposure to air or water. Its reactivity is both an advantage and a limitation, depending on how it is used in industrial processes.
Synthesis of Ethylene Sulfate
The production of ethylene sulfate involves controlled chemical reactions in laboratory or industrial settings. It is generally synthesized through reactions involving ethylene derivatives and sulfate-based reagents under specific temperature and pressure conditions.
The synthesis process requires careful monitoring because improper conditions can lead to unwanted side reactions or reduced yield. Chemists focus on optimizing reaction conditions to ensure that the final product is pure and stable. This makes its production a specialized process that is usually carried out in advanced chemical manufacturing facilities.
Industrial Uses of Ethylene Sulfate
Ethylene sulfate is primarily used as an intermediate in chemical synthesis. It plays an important role in the production of various organic compounds that are later used in pharmaceuticals, materials science, and industrial chemistry.
In some cases, it is used in research laboratories to study reaction mechanisms and develop new chemical pathways. Its ability to undergo controlled reactions makes it valuable for experimental chemistry.
In industrial applications, ethylene sulfate contributes to the development of compounds that may be used in coatings, polymers, and other advanced materials. Its versatility allows it to be adapted for different chemical processes depending on the requirements of the industry.
Role in Organic Chemistry
In organic chemistry, ethylene sulfate is considered an important reactive intermediate. It is often used to facilitate the formation of more complex molecular structures. Chemists use it to study reaction mechanisms and understand how different chemical bonds are formed or broken.
Its role in ring-opening reactions is particularly significant because it allows the transformation of simple structures into more complex ones. This makes it a valuable tool in synthetic organic chemistry, where precision and control are essential.
Safety Considerations
Handling ethylene sulfate requires strict safety measures due to its reactive nature. It should only be used in well-equipped laboratories with proper ventilation and protective equipment. Direct exposure can pose risks, so it is handled with caution by trained professionals.
Storage conditions are also important. It must be kept in tightly sealed containers and away from moisture and heat sources. Safety protocols are established to ensure that accidental reactions do not occur during handling or transportation.
Workers dealing with ethylene sulfate typically follow standardized chemical safety guidelines, including the use of gloves, goggles, and protective clothing. Proper training is essential to minimize risks associated with its use.
Environmental Impact
Like many industrial chemicals, ethylene sulfate must be managed carefully to prevent environmental contamination. Improper disposal can lead to chemical reactions in the environment, which may affect soil and water quality.
Industrial facilities that use ethylene sulfate are required to follow environmental regulations to ensure safe disposal and minimize ecological impact. Treatment processes are often used to neutralize or safely break down chemical residues before disposal.
Sustainable chemical management practices are becoming increasingly important, and compounds like ethylene sulfate are handled with greater responsibility to reduce environmental risks.
Research and Scientific Importance
Ethylene sulfate continues to be a subject of research in modern chemistry. Scientists study its behavior in different reaction environments to better understand its potential applications. Research focuses on improving synthesis methods, increasing efficiency, and exploring new industrial uses.
Its role in developing new materials and chemical processes makes it relevant in advanced scientific studies. As chemistry continues to evolve, compounds like ethylene sulfate remain important building blocks in innovation.
Conclusion
Ethylene sulfate is a chemically significant compound with important applications in industrial and research chemistry. Its unique structure and reactivity make it valuable in organic synthesis and material development. However, due to its reactive nature, it must be handled with care and under strict safety conditions.
From its synthesis to its industrial applications, ethylene sulfate plays a crucial role in modern chemical processes. Ongoing research continues to expand its potential uses, making it an important compound in the field of chemistry.
- Business
- Technology
- Finance
- Health
- Fashion
- Lifestyle
- Travel
- Food
- Educação
- Real Estate
- Automobile
- Entertainment
- Sports
- Pets
- Home Decor
- Gardening
- Parenting
- Wedding
- Beauty
- Gaming
- Photography
- Music
- Movies
- News
- Politics
- Religion
- Astrology
- Law
- Insurance
- Jobs
- Freelancing
- Remote Work
- Blogging
- E-commerce
- Endereço Web
- OpenCart
- Social Media
- Graphic Design
- Web Design
- Programming
- Mobile Apps
- Artificial Intelligence
- SaaS
- Cyber Security
- Cloud Computing
- Hosting
- SEO
- Content Writing
- Email Marketing
- Affiliate Marketing
- YouTube
- Podcasting
- Interior Design
- DIY
- Crafts
- Art
- Architecture
- Environment
- Agriculture
- Animals
- Fishing
- Hunting
- Survival
- Outdoor
- Fitness
- Yoga
- Meditation
- Mental Health
- Skin Care
- Hair Care
- Makeup
- Jewelry
- Sarees
- Salwar Kameez
- Lehenga Choli
- Kurtis
- Men Fashion
- Women Fashion
- Kids Fashion
- Footwear
- Bags
- Watches
- Luxury
- Shopping
- Coupons
- Electronics
- Smartphones
- Laptops
- Tablets
- Cameras
- Home Appliances
- Kitchen
- Furniture
- Cleaning
- Baby Care
- Senior Care
- Relationships
- Self Improvement
- Motivation
- Quotes
- Festivals
- Eventos
- Careers
- Remote Work
- Productivity
- Wholesale
- Manufacturing
- Import Export
- Logistics
- Supply Chain
- Human Resources
- Customer Service
